UTI Vaccine
Struggling with recurrent UTIs that keep coming back despite antibiotics? A UTI vaccine may offer a long-term solution by helping your body prevent infections before they start.
UTI Vaccine: Treatment for recurrent urinary tract infections
The UTI vaccine is a pioneering preventative treatment designed to reduce the frequency of recurrent urinary tract infections by training the immune system to naturally recognise and fight the bacteria that commonly cause UTIs.
Unlike antibiotics, which treat infections after they occur, this vaccine works proactively to help prevent future infections.
This may offer those who are susceptible to UTIs:
- Reduced reliance on antibiotics
- Higher prevention of recurrence rates
- Longer term protection from infection
- An improved quality of life free from debilitating symptoms
We provide a UTI vaccine, taken as an under the tongue (sublingual) spray, on a named patient basis at The Urology Partnership.
Call 0118 920 7040 to book a consultation and check your eligibility.
Can a vaccine prevent recurrent UTIs?
Clinical research shows that UTI vaccines can significantly reduce infection frequency, particularly in patients who have not responded to conventional treatments.
The Urology Partnership holds the longest-standing experience with UTI vaccines in the United Kingdom. Pioneering this treatment, our Consultant Urologists Mr Bob Yang and Mr Steve Foley were the first to introduce it to the UK and served as leads for the international randomised control trials in the UK.
Our studies suggest:
- Up to 70–80% reduction in UTIs within the first year (Yang and Foley, 2018)
- Over 50% of patients remain infection-free long term (Lorenzo-Gómez et al., 2022)
- Protection may last several years in some patients (Lorenzo-Gómez et al., 2022)
Find out more about our UTI vaccine clinical research and trials.
Why consider a UTI vaccine instead of antibiotics?
Many patients with recurrent UTIs often rely on repeated and prolonged courses of antibiotics. While effective in the short term, this approach has limitations.
UTI Vaccine vs Antibiotics
| Feature | Antibiotics | Vaccine |
|---|---|---|
| Purpose | Treat active infection | Prevent future infections |
| Duration | Short-term protection | Long-term protection |
| Resistance risk | High | No known resistance |
| Microbiome impact | Can be disruptive | Minimal impact |
With rising antibiotic resistance, alternative approaches to prevention are also becoming increasingly important.
The World Health Organisation warns that antimicrobial resistance is undermining the effectiveness of life-saving treatments and increasing the risk from common infections. (World Health Organization, 2025)
The science behind the use of vaccine/immunostimulation in patients with recurrent infections won a Nobel Prize for medicine in 2011. (Nobel Prize Outreach, 2011)
Various types of UTI vaccines are now being developed for treating recurrent UTIs including ones that can be swallowed, injected, applied to the vagina and sprayed under the tongue (sub-lingual). All work by boosting the body’s natural immune system to fight off a UTI before it can take hold.
How does the UTI vaccine work?
The vaccine contains inactivated forms of the most common bacteria responsible for UTIs, including:
- Escherichia coli
- Klebsiella pneumoniae
- Proteus vulgaris
- Enterococcus faecalis
When administered, it stimulates the immune system to recognise and respond more effectively to these bacteria, helping prevent infection before it develops.
How is the UTI vaccine taken?
The treatment is simple and non-invasive:
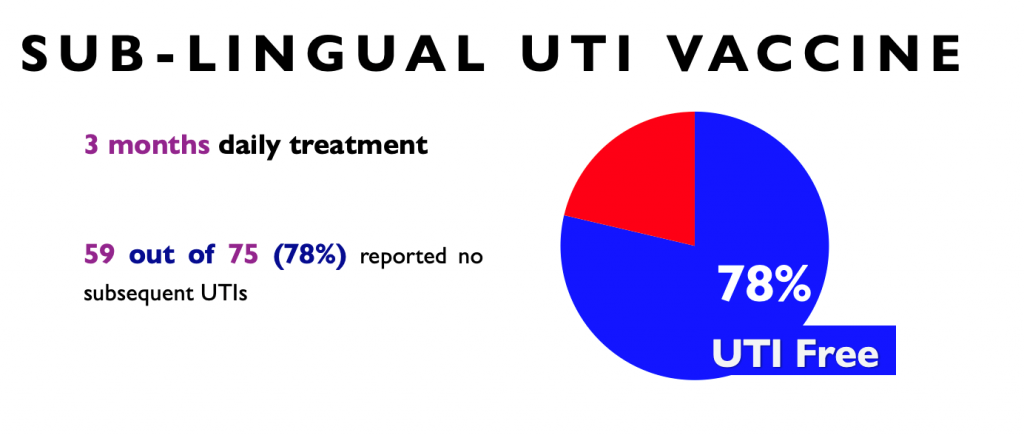
- Administered as a sublingual (under-the-tongue) pineapple flavoured spray
- Taken daily for 3 months
- No injections required
Is the UTI vaccine available in the UK?
UTI vaccines are currently unlicensed in the UK and are not available on the NHS or via a pharmacy.
However, they may be accessed privately on a named patient basis following a specialist consultation.
Is it safe if the vaccine is unlicensed?
Yes – when prescribed appropriately.
“Unlicensed” means the treatment is still undergoing regulatory approval, not that it is unsafe. It can be legally prescribed by specialists where there is sufficient clinical evidence and a clear patient need.
At our clinic, treatment is:
- Prescribed by experienced consultant urologists
- Based on clinical evidence and research
- Monitored closely for safety and effectiveness
Who is eligible for a UTI vaccine?
The treatment is typically considered for:
- Adults with recurrent UTIs
- Patients who have not responded to preventive antibiotics
- Those seeking to reduce long-term antibiotic use
A consultation is required to assess suitability.
Call 0118 920 7040 to schedule an appointment with one of our urology specialists.
How effective is the UTI vaccine?
Our clinical research and experience show promising results in efficacy, safety and reduction in UTI recurrence rates for men and women who had not responded to standard treatments, including antibiotic prophylaxis.
Key findings:
- 70–80% reduction in infections within 12 months (Yang and Foley, 2018)
- 54% of patients remained UTI-free for up to 9 years (Lorenzo-Gómez et al., 2022)
- Strong safety profile with minimal side effects (Lorenzo-Gómez et al., 2022)
These outcomes are particularly significant for patients who had previously exhausted other treatment options.
Our studies in the news:
- Game changer UTI vaccine stops infection for nine years – The Telegraph, 6 April 2024
- Ground breaking UTI vaccine could stop infection for nine years – The Independent, 7 April 2024
- Vaccine that can stop UTIs for nine years – a drug that could prove a saviour for up to 1.7m Britons – The Daily Mail, 7 April 2024
Where can you get a UTI vaccine in the UK?
The Urology Partnership offers access to UTI vaccine treatment through private consultation.
Please call 0118 920 7040 to book your appointment.
Why choose The Urology Partnership?
We are one of the most experienced centres in the UK for UTI vaccine treatment.
- Over 10 years of clinical experience
- Among the first to introduce this treatment in the UK
- Active involvement in international clinical research
- Published in leading peer-reviewed journals
Our consultants combine research expertise with personalised patient care.
Take the first step towards preventing recurrent UTIs
If you’re tired of repeated infections and ongoing antibiotics, a UTI vaccine may offer a new approach to long-term prevention.
Call now to arrange your consultation: 0118 920 7040
UTI Vaccine FAQs
There are very few side effects. Over the last 10 years of experience, 90% of patients have tolerated the treatment with no adverse reactions whatsoever. For the small number who do experience side effects, the vast majority are mild and self-limiting. If you do develop any side effects, please inform your consultant so we can monitor your progress and take appropriate action.
Not entirely; it serves a different purpose. If you experience an acute flare-up, antibiotics are still required to treat the active infection. However, the UTI vaccine is a preventative measure designed to significantly reduce the frequency of your UTIs, thereby decreasing the overall volume of antibiotics you will need over time.
Generally, we find that the primary protection lasts for 1 to 2 years before a top-up dose is required. In the longer term, however, the protective benefits can endure for up to 9 years, and potentially even longer.
The current standard in many clinics is to prescribe antibiotics, at either high or low doses, for a prolonged period.
However, research – which our consultants at the Urology Partnership are actively involved in – shows that long-term antibiotic use has a substantial side-effect profile, particularly concerning damage to the healthy gut microbiome.
Our philosophy is to avoid generalised, long-term antibiotics wherever possible, favouring targeted, focused antibiotics or non-antibiotic preventative methods instead.
The regulatory process for medicines in the UK is both robust and thorough. Whilst we cannot predict how long this process will take, we will continue to provide the manufacturers and their UK partners the clinical evidence in our research to facilitate the application and we hope to bring this treatment to all patients in the NHS in the future.
Clinical References
- Nobel Prize Outreach (2011) The Nobel Prize in Physiology or Medicine 2011. Available at: https://www.nobelprize.org/prizes/medicine/2011/summary/ (Accessed: 18 March 2026)
- Nobel Prize Outreach (2011) Advanced information: The Nobel Prize in Physiology or Medicine 2011. Available at: https://www.nobelprize.org/prizes/medicine/2011/advanced-information/ (Accessed: 18 March 2026).
- World Health Organization (2025) Global antibiotic resistance surveillance report 2025. Available at: https://www.who.int/publications/i/item/9789240116337/ (Accessed: 30 March 2026).
- Bob Yang and Steve Foley (2018) ‘First experience in the UK of treating women with recurrent urinary tract infections with a bacterial vaccine’, BJU International, 121(2), pp. 289–292. Available at: https://doi.org/10.1111/bju.14067
- Maria-Fernanda Lorenzo-Gómez, Steve Foley, J Curtis Nickel, García-Cenador, M.-B., Padilla-Fernández, B.-Y., González-Casado, I., Martínez-Huélamo, M., Bob Yang, Blick, C., Ferreira, F., Caballero, R., Saz-Leal, P. and Casanovas, M. (2022) ‘Sublingual MV140 for prevention of recurrent urinary tract infections’, NEJM Evidence, 1(4). Available at: https://doi.org/10.1056/EVIDoa2100018
Next Steps
Our UTI Vaccine Specialists
Get fast access to leading specialists for the swift diagnosis and treatment of urological conditions in a private clinic environment.
If you would like more information or wish to arrange a consultation with one of our specialist consultant urological surgeons then please either Call 0118 920 7040 or complete the form below.
Insured patients
Contact your GP and ask for a referral to the Urology Partnership.
All consultations, investigations and treatments are covered by major insurance companies (depending on policy).
Funding your own treatment
Self-funding initial consultation fee is £250. Follow up fees are £175.
Consultation charges are exclusive of any tests and other investigations that the consultant may wish to carry out.
Reviewed by
Reviewed by: Mr Bob Yang, Consultant Urological Surgeon
Date published: 24 January 2020
Date modified: 30 March 2026
Last reviewed: 27 March 2026